Tell us about the biosynthetic “brains” you’re making.
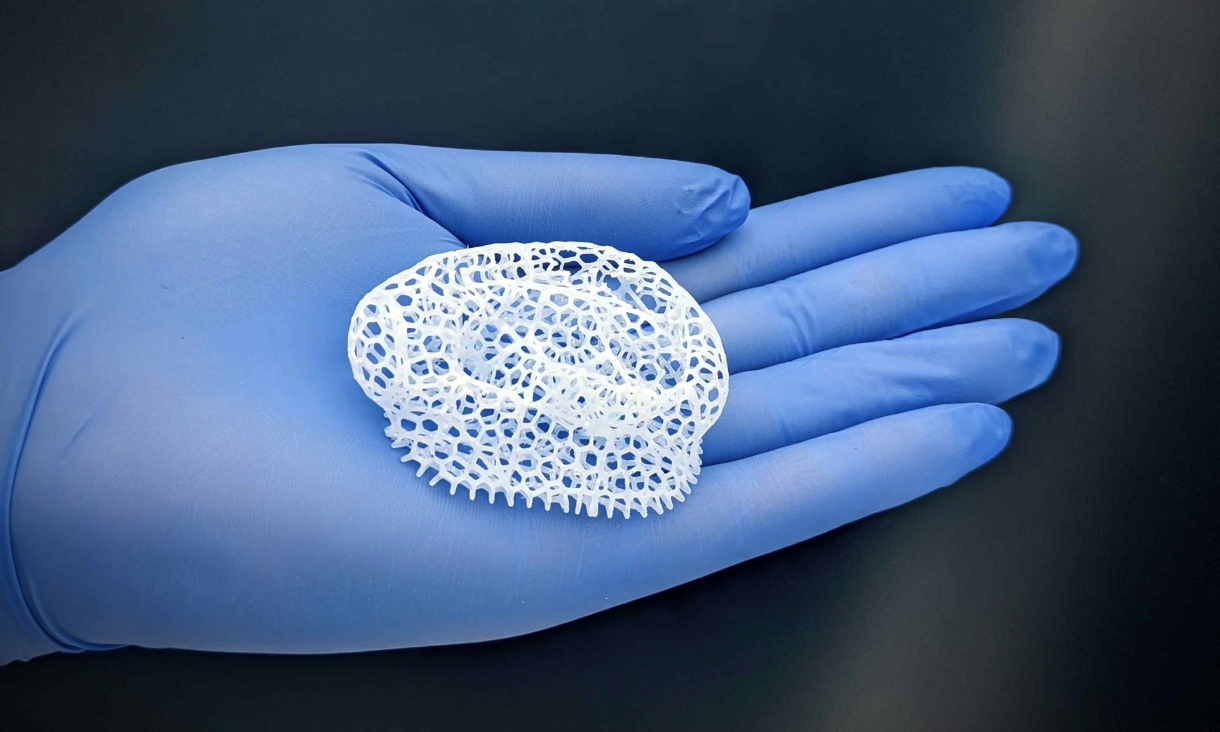
When you’re trying to understand how the brain works, and how to fix it when it goes wrong, looking at cells on two-dimensional slides only takes you so far. So we build in three dimensions, using 3D bioprinting.
The ‘brains’ we make are tiny, around 3mm by 3mm, but there is enough functioning brain there to be studied and analysed (and besides, the human brain cortex is only 3mm thick).
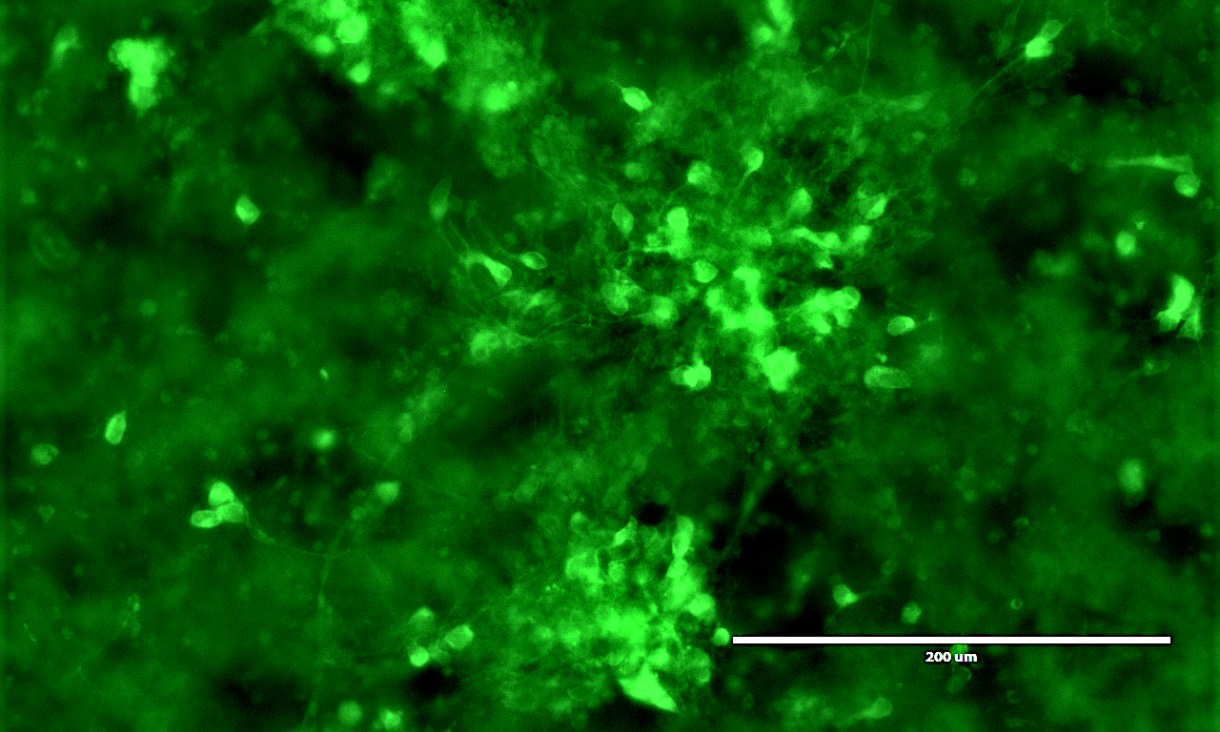
Our little brain blocks are made from skin cells, which we reprogram into stem cells that can make neurons. The block of ‘brain’ is suspended in a 3D collagen matrix and put on array of electrodes.
Using electrical stimulation or adding neurochemicals, we can get the neurons to fire. The electrode array records that activity – which mimics the firing of the nervous system – to give us an in-depth picture of what is happening.
Essentially these are personalised synthetic brain constructs, ideal for scientists working to understand and treat neurological conditions.
How are these “brains” helping us with research into epilepsy?
Epilepsy affects 1 in 100 people, but we know that about half of those develop the condition later in life, after experiencing some injury to the brain when they were younger.
We think that may be because some people have a genetic predisposition, but right now we can’t predict who may be at risk of developing epilepsy because of head injury.
In this research, we’re collaborating with the neurologists at the Murdoch Children’s Research Institute who discovered a genetic mutation that causes epilepsy, as well as neurologists at St Vincent’s Hospital Melbourne.
We take skin cells from people who have that genetic mutation, remove the mutation and grow biosynthetic ‘brain’ from those genetically-edited cells. For comparison, we also make brain out of their ‘epilepsy-positive’, unedited cells.
We then test the ‘brains’ we make from these cells by inducing a certain level of injury and seeing if – and when – they display epileptic-like activity.
What we’re ultimately aiming for is a simple genetic test that could determine if you are likely to develop epilepsy from minor head trauma, such as through playing AFL football or other sports.