In this project we apply advanced computer modelling techniques to investigate the role of surfaces and nanoparticles in amyloid fibril formation.
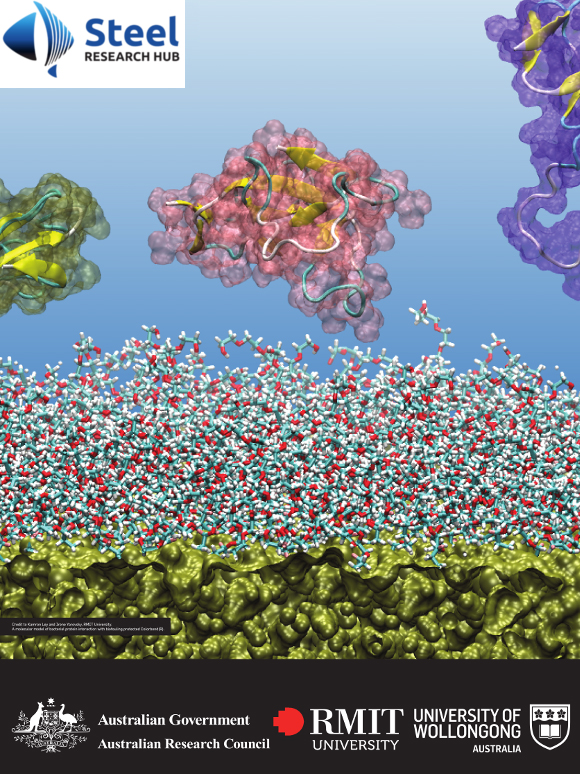
One of the greatest challenges currently posed by the industrial uptake of nanomaterials is the lack of understanding of the interactions of these novel molecular systems with biological environments. Nanomaterials present a high surface area that can interact with proteins and promote a locally increased protein concentration leading to aggregation. A specific type of protein aggregation caused by the partial misfolding and self-association of proteins to form insoluble amyloid fibrils occurs in a number of common and debilitating diseases, including Alzheimer’s and Parkinson disease. We have been studying amyloid fibril formation by human apolipoprotein (ApoC-II) as a model to explore the factors that affect the self-assembly process.
Outcomes of the work include a molecular level understanding of protein misfolding related to disease, a rational basis for designing inhibitors to protein aggregation and the establishment of design principles for novel nanomaterials via controlled self assembly of proteins and peptides on surfaces.
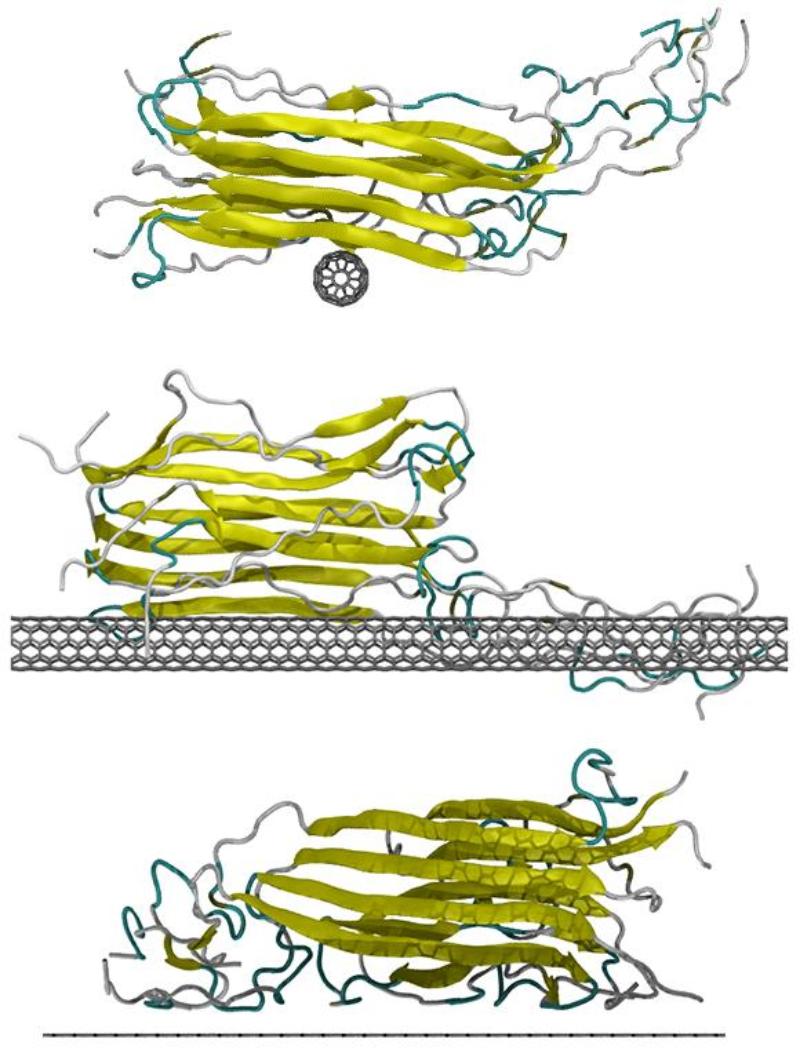 Atomistic model of an amyloid fibril interacting with a fullerene, nanotube and graphene surfaces.
Atomistic model of an amyloid fibril interacting with a fullerene, nanotube and graphene surfaces.
Key people
- Professor Irene Yarovsky, Chief Investigator and team leader
- Nevena Todorova, Research Fellow
- Dr Andrew Hung, Research Fellow
- Adam Makarucha, PhD student (completed 2015)
Partners
- Associate Professor Geoff Howlett, Bio21 Institute, University of Melbourne
- Dr Michael Griffin, Bio21 Institute, University of Melbourne
- Dr Arash A. Mostofi, Department of Materials and Physics, Imperial College London UK
- Y. Mao, N. Todorova, C. O. Zlatic, P. R. Gooley, M. D. W. Griffin, G. J. Howlett, I. Yarovsky “Solution Conditions Affect the Ability of the K30D Mutation To Prevent Amyloid Fibril Formation by Apolipoprotein C-II: Insights from Experiments and Theoretical Simulations” Biochemistry, 55 (27), pp 3815–3824 (2016)
- Y. Mao, C. Zlatic, M. D. W. Griffin, G. J. Howlett, N. Todorova, I. Yarovsky and P. R. Gooley, “Hydrogen/deuterium exchange and molecular dynamics analysis of amyloid fibrils formed by a D69K charge-pair mutant of human apolipoprotein C-II” Biochemistry, 54 (31), pp 4805–4814 (2015)
- M. D. W. Griffin, L. Yeung, A. Hung, N. Todorova, Y.F Mok, J.A. Karas, P. R. Gooley, I. Yarovsky, G. J. Howlett "Cyclic peptide inhibitor of apoC-II peptide fibril formation: mechanistic insight from NMR and molecular dynamics analysis" J. Mol. Biol. 416, pp. 642-655 (2012)
- N. Todorova, A. J. Makarucha, N. Hine, A. Mostofi, I. Yarovsky, “Dimensionality of carbon nanomaterials determines the aggregation propensity of amyloidogenic peptides“ PLoS Comp Biology, 9(12): e1003360, (2013)
- N. Todorova, L. Yeung, A. Hung, I. Yarovsky, “Janus Cyclic Peptides: A New Approach to Amyloid Fibril Inhibition?“ PLoS ONE, Vol. 8(2) e57437 (2013)
- C. L. Teoh, C. L. L. Pham, N. Todorova, A. Hung, C. N. Lincoln, E. Lees, Y. H. Lam, K. J. Binger, N. H. Thomson, S. E. Radford, T. A. Smith, S. A. Müller, A. Engel, M. D . W. Griffin, I. Yarovsky, P. R. Gooley and G. J. Howlett “A structural model of apolipoprotein C-II amyloid fibrils: Experimental characterization and molecular dynamics simulations“, J. Mol. Biol. Vol. 405, pp. 1246-1266 (2011)
- A. J. Makarucha, N. Todorova, I. Yarovsky “Nanomaterials in biological environment: a review of computer modelling studies“ Eur. Biophys. J, Vol. 40, pp.103-115 (2011)
- N. Todorova, A. Hung, S.M. Maaser, M.D .W. Griffin, J. Karas, G.J. Howlett, I. Yarovsky “Effects of mutation on the amyloidogenic propensity of apolipoprotein C-II 60-70 peptide“Phys. Chem. Chem. Phys. Vol. 12, pp. 14762, (2010)
- N. Todorova, I. Yarovsky "Molecular modelling of peptide folding, misfolding and aggregation phenomena.", International Conference on Computational Science, ICCS 2010, Amsterdam, Procedia Computer Science, Vol. 1, pp. 1179, (2010)
- N. Todorova, A. Hung, I. Yarovsky, "Lipid concentration effects on the amyloidogenic apoC-II 60-70 peptide: A computational study." J. Phys. Chem. B, Vol. 114, pp. 7974, (2010)
- N. Todorova, A. Hung, I. Yarovsky, “Application of computational modelling to protein folding and aggregation studies“ Proceedings of the 10th WSEAS International Conference on Mathematics and Computers in Biology and Chemistry, Czech Republic (2009) ISBN: 978-960-474-062-8
- A. Hung, N. Todorova, I. Yarovsky, “Computer simulation studies of abnormal protein aggregation.“ Proceedings of the 2nd WSEAS International Conference on Biomedical Electronics and Biomedical Informatics, Russia (2009) ISBN: 978-960-474-110-6
- A. Hung, M.D.W. Griffin, G. J. Howlett, I. Yarovsky, “Lipids enhance amyloidogenic peptide oligomerisation but inhibit fibril formation“ J. Phys. Chem. B, Vol. 113, pp. 9447 (2009)
- A. Hung, M.D.W. Griffin, G.J. Howlett, I. Yarovsky, “Effects of oxidation, pH and lipids on amyloidogenic peptide structure: Implications for fibril formation?“ Eur. Biophys. J, Vol. 38, pp. 99 (2008)


